ԭ��BG���Ź���
��Ʒ����
���Ŷ�̬
��ϵ����
������ ���� 023-61213011
֤ȯ�� ���� 023-61213003
�ʱ� 401121
��ַ �������山������·2��
|
��Ʒ���� |
Ӣ���� | CAS NO. | �ṹʽ Structure | Remark & Status | �Ǽ�״̬ |
| ���������ƽ | Quetiapine Fumarate | 111974-72-2 | 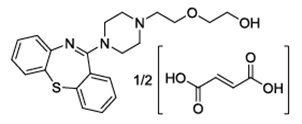 |
GMP DMF |
A |
| ����� | Rocuronium bromide | 119302-91-9 | 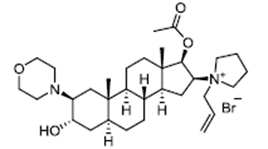 |
GMP DMF |
A |
| �Ŷ��������� | L-ornithine L-aspartate | 3230-94-2 | 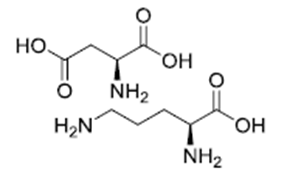 |
GMP DMF Commercial |
A |
| ͪ��������� | α-Ketophenylalanine Calcium | 51828-93-4 | 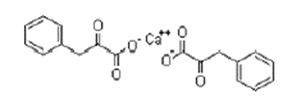 |
GMP Commercial |
A |
| ͪ�Ӱ���� | Calcium alpha-ketovaline | 51828-94-5 | 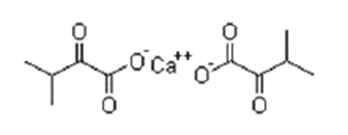 |
GMP Commercial |
A |
| ͪ������� | α-Ketoleucine Calcium | 51828-95-6 | 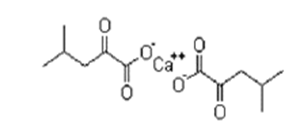 |
GMP Commercial |
A |
| ����ͪ��������� | D,L-α-Ketoisoleucine Calcium | 66872-75-1 | 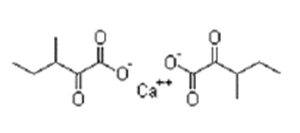 |
GMP Commercial |
A |
| �����ǵ������ | D,L-α-Hydroxymethionine Calcium | 4857-44-7 | 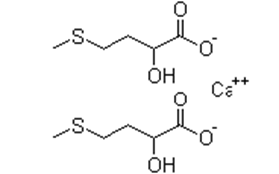 |
GMP Commercial |
A |
| ��������� | Hydroxychloroquine sulfate | 747-36-4 | 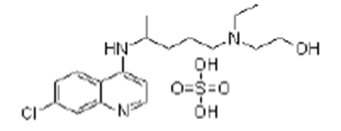 |
GMP Commercial |
A |
| ��˾���������� | Esomeprazole Sodium | 161796-78-7 | 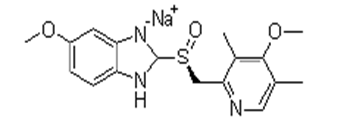 |
GMP Commercial |
A |
| ������Īɳ���� | Mosapride Citrate | 112885-42-4 | 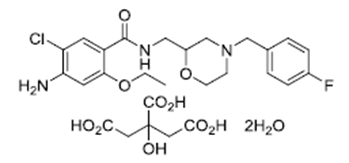 |
GMP DMF |
A |
| ������̹ | Oxiracetam | 62613-82-5 | 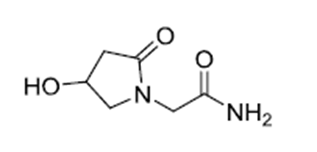 |
GMP Commercial |
A |
| ������� | Rebamipide | 90098-04-7 | 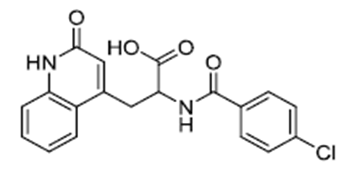 |
GMP DMF |
A |
| �Dz�˾�� | Febuxostat | 144060-53-7 | 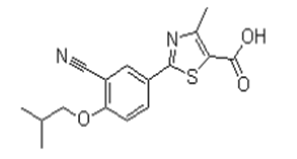 |
GMP Commercial |
A |
| �����ֿ���ƽ | Lercanidipine hydrochloride | 132866-11-6 | 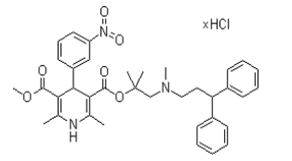 |
GMP DMF |
A |
| ������� | Nicergoline | 27848-84-6 | 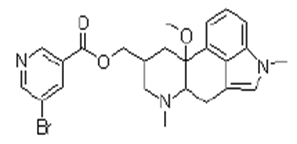 |
GMP DMF Korea MFDS |
A |
| ��ӻ��� | Tigecycline | 220620-09-7 | 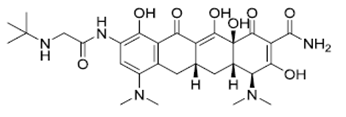 |
GMP USDMF#27477 |
A |
| ����ͷ���� | Latamoxef Sodium | 64953-12-4 | 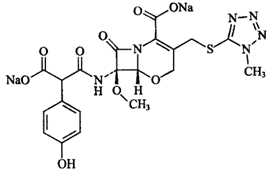 |
GMP Commercial |
A |
| ͷ�������� | Cefmetazole Sodium | 56796-39-5 | 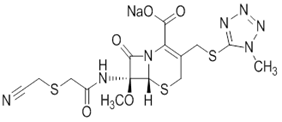 |
GMP DMF |
A |
| ͷ������� | Ceftizoxime sodium | 68401-82-1 | 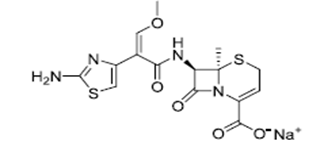 |
GMP DMF |
A |
| ������ | Aztreonam | 78110-38-0 | 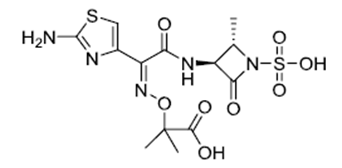 |
GMP DMF |
A |
| ������/������ | Aztreonam And Arginine | ---- | 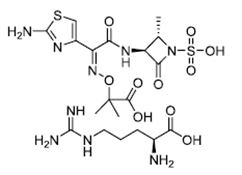 |
GMP DMF |
A |
| ����ù�������� | Clindamycin phosphate | 24729-96-2 | 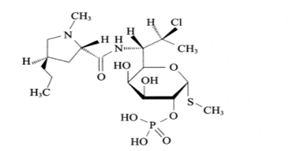 |
GMP Commercial |
A |
| �������� | Voriconazole | 137234-62-9 | 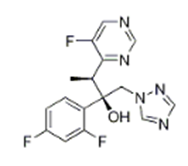 |
GMP DMF |
A |
| ͷ������ƥ�� | Cefditoren pivoxil | 117467-28-4 | 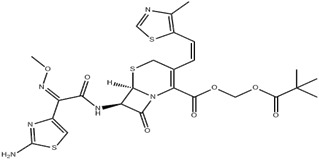 |
GMP DMF |
A |
| ���������� | Oxacillin Sodium | 1173-88-2 | 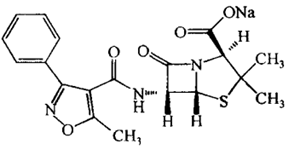 |
GMP DMF |
A |
| �����о� | Empagliflozin | 864070-44-0 | 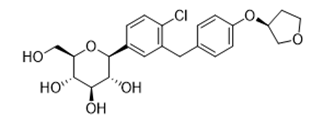 |
GMP Commercial |
A |
| �����ض� | Piribedil | 3605-1-4 | 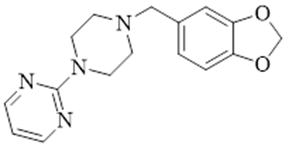 |
GMP | A |
| ����ͷ���氲 | Cefotiam Hydrochloride | 66309-69-1 | 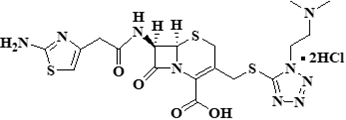 |
GMP Commercial |
A |
| ����� | Indobufen | 63610-08-2 | 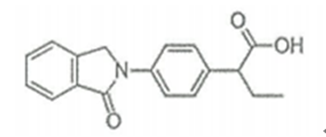 |
GMP | A |
| �ױ���Ӣ�� | Fosphenytoin sodium | 92134-98-0 | 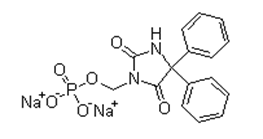 |
GMP DMF USDMF#32816 |
A |
| ������ | Aztreonam | 78110-38-0 | 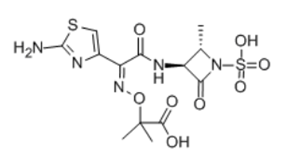 |
DMF USDMF#27508 |
A |
| ������ | Alpha Lipoic Acid | 1077-28-7 | 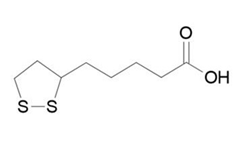 |
Y20210000898 | A |
| ���Ἢ������ | Gemcitabine Hydrochloride | 122111-03-9 | 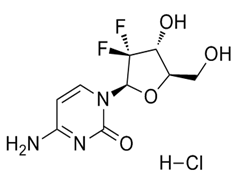 |
GMP/ DMF/ CEP R1-CEP 2009-042-Rev01 Indian DCGI RC-BD-002278 Korea MFDS Commercial |
A |
| �������˾�� | Granisetron Hydrochloride | 107007-99-8 | 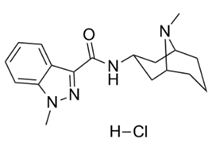 |
GMP Commercial |
A |
| �����˾�� | Ondansetron Hydrochloride |
103639-04-9 | 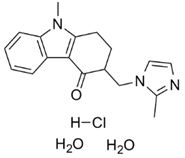 |
GMP Commercial |
A |
| ������� | Doxofylline | 69975-86-6 | 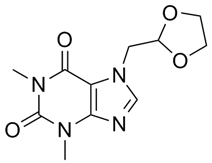 |
GMP Commercial |
A |
| ������ | Carvedilol | 72956-09-03 | 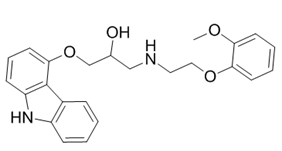 |
GMP Commercial |
A |
| ������ | Sulindac | 38194-50-2 | 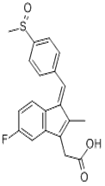 |
GMP Commercial |
A |
| ������������ | Toremifene Citrate |
89778-27-8 | 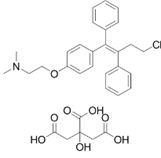 |
GMP/ PMDA Commercial |
A |
| �������� | Flumazenil | 78755-81-4 | 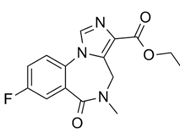 |
GMP Commercial |
A |
| �����ḣĪ���� | Formoterol Fumarate |
43229-80-7 | 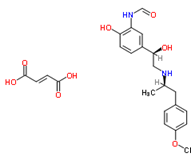 |
GMP Commercial |
A |
| ������������� | Levocetirizine Hydrochloride |
130018-87-0 | 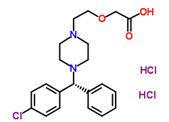 |
GMP Commercial |
A |
| ���ȷ� | Baclofen | 1134-47-0 | 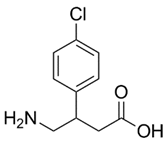 |
GMP Commercial |
A |
| ������ | Regorafenib | 755037-03-7 | 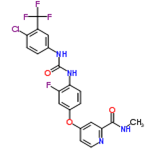 |
Commercial | A |
| ������Τ | Peramivir Trihydrate | 1041434-82-5 | 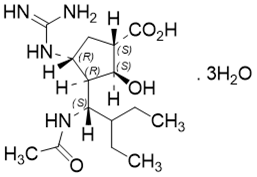 |
Commercial | A |
| ���������� | Pamidronate disodium |
109552-15-2 | 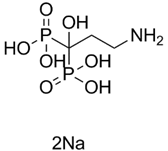 |
GMP Commercial |
A |
| �������з��沼 | Tofacitinib citrate | 540737-29-9 | 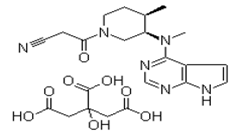 |
Commercial | A |
| �������ù�� | Gentamicin Sulfate | 1405-41-0 | 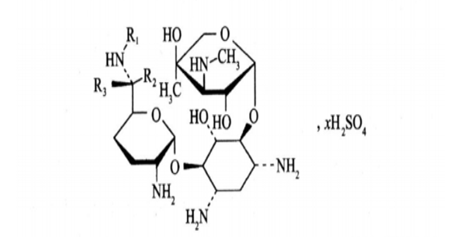 |
GMP DMF CEP Indian DCGI Korea MFDS Commercial |
A |




